Excellent alternative to autogenous and allogenous bone.
The mineralized inorganic bone matrix of the Bonefill has a porous macro and micro structure similar to the human cortical and spongy bones. In granulate form, Bonefill Dense, Porous & Mix act as osteoconductive mechanism promoting bone growth and regeneration. With time, the Bonefill is partially remodeled through the action of osteoclasts and osteoblasts, being a viable alternative to autologous bone in defects suitable for its use and indication.
The highly purified osteoconductive mineral structure is produced from natural bone through a multiphase process, complying with the safety regulations established by the control agencies. The fresh bone is crushed, receiving a sequence of baths that solubilize the organic structures such as, for example, remaining cells, fibers and proteins, with only the mineral portion remaining this way in order to avoid the induction of possible immunogenic processes in the body.
The products made of mineralized bovine bone have an expected incorporation of 6 to 9 months.
Bonefill are extensively used in medicine and dentistry.
Due to the natural origin, Bonefill is comparable to the mineral and morphological structure of the mineralized human bone, it is biocompatible, does not present cytotoxicity, acute systemic toxicity, carcinogenicity and it is not a sensitizing product [ISO 10993-1].
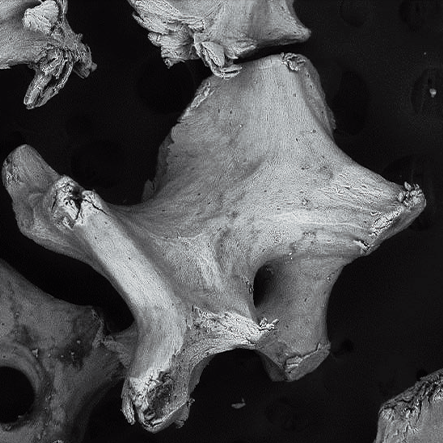
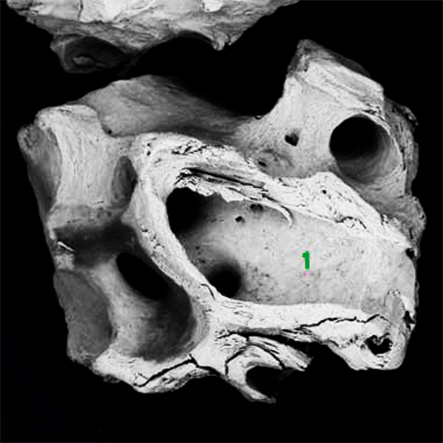
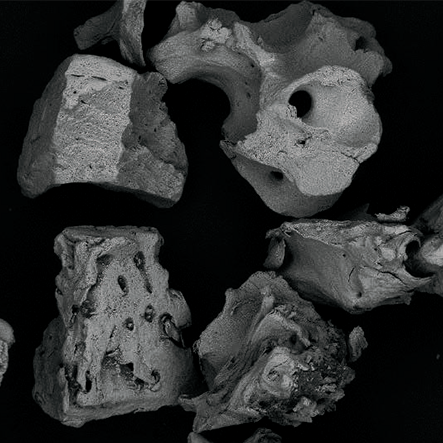
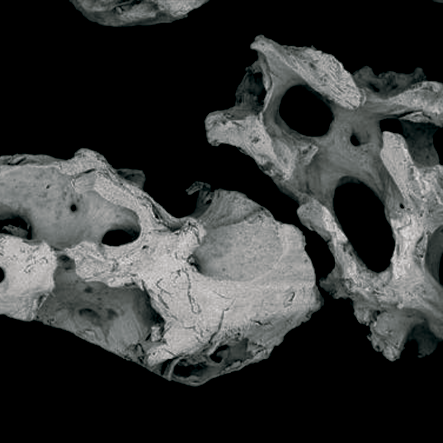
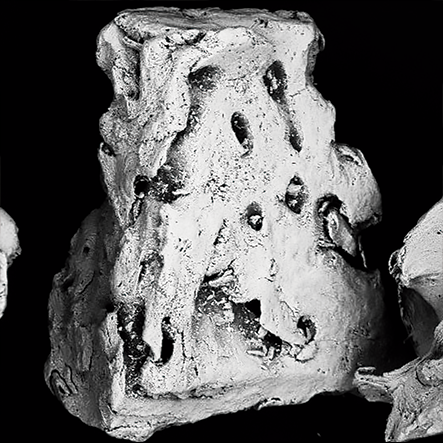
Topography.
The surface helps the absorption of proteins in the Bonefill Porous particles, enabling the efficient adhesion of the osteoblasts. This biological interaction enables a reliable bone formation.
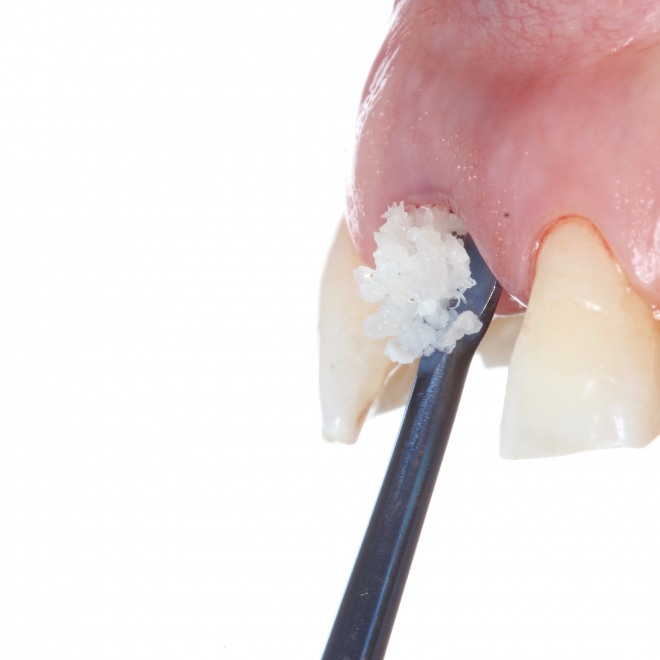
bonefill.porous granules
bonefill.mix granules
bonefill.denso granules
Hydrophilicity.
The rapid and complete hydration with blood or saline solution is an important feature of the handling, new bone formation and clinical success. Its strong capillary action allows the rapid and efficient penetration of particles with fluids in the material, nutrients and blood through its three-dimensional network of the trabecular bone, resulting in excellent handling, application and predictability in daily clinical use.
"
Purified
Bonefill is subjected to a multiphase purification system that removes the organic material content from the bone. This process results in a Bonefill chemically and structurally similar to mineralized human bone (natural nanocrystalline apatite). Furthermore, it proved that Bonefill is biocompatible.
"
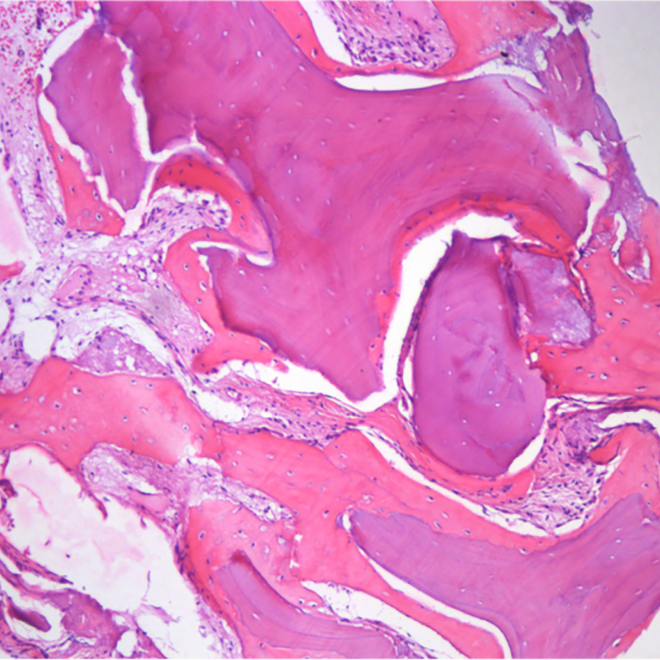
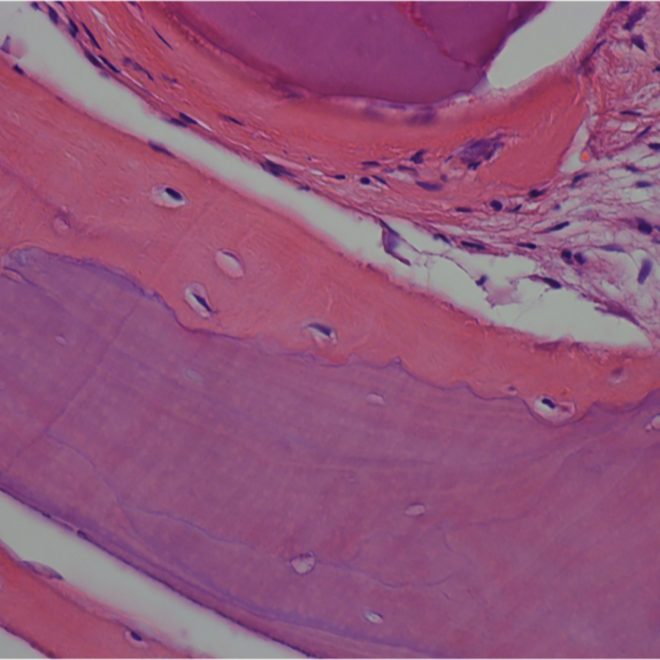
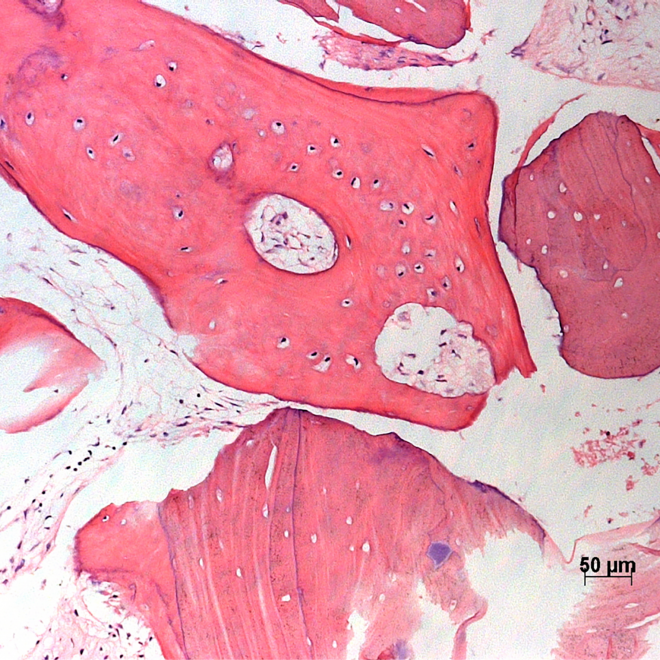
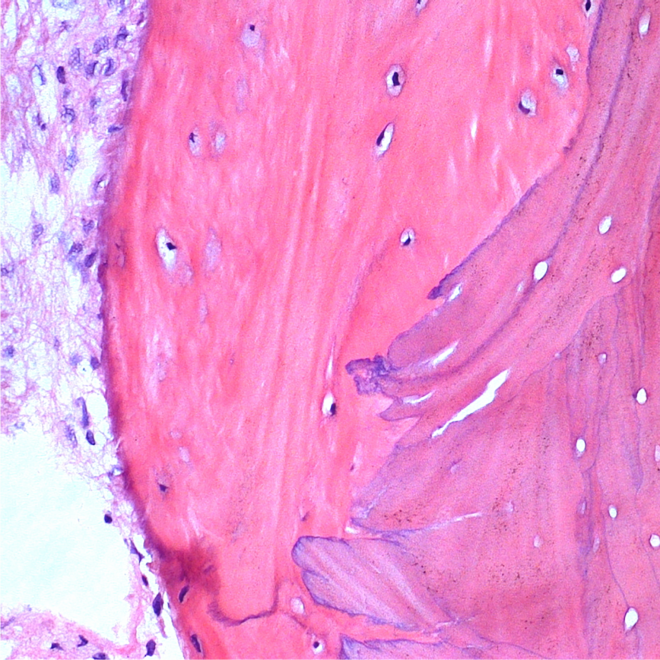
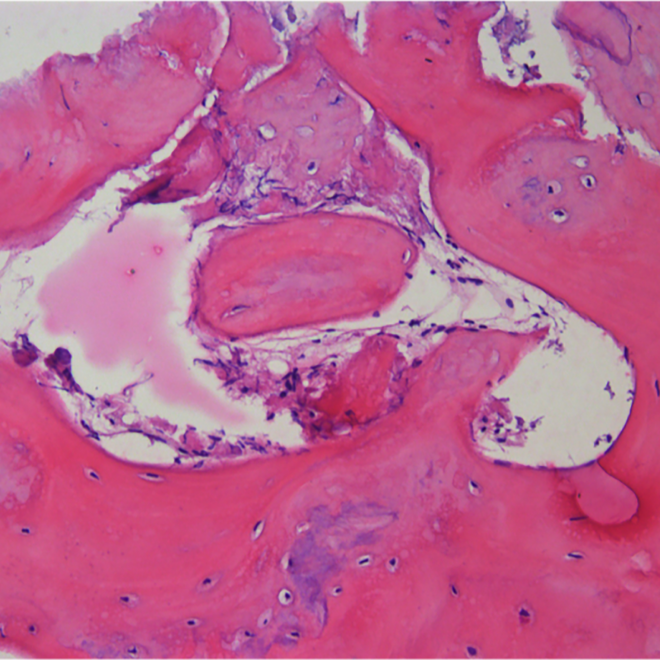
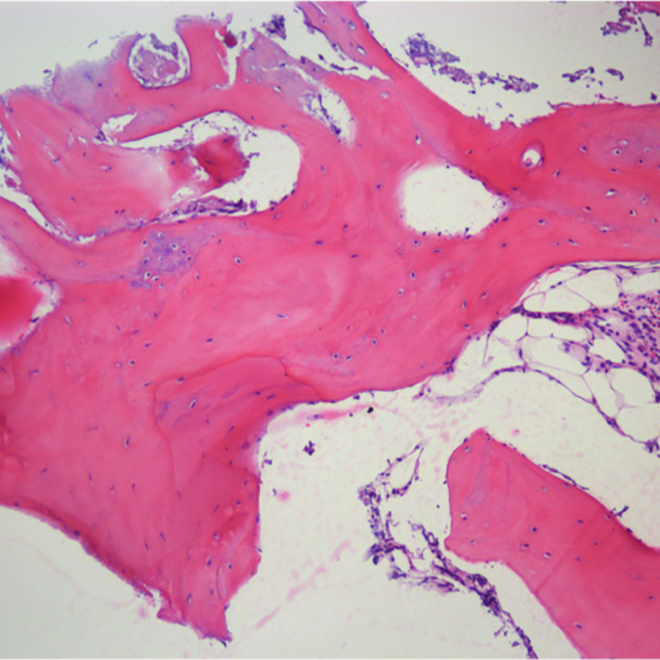
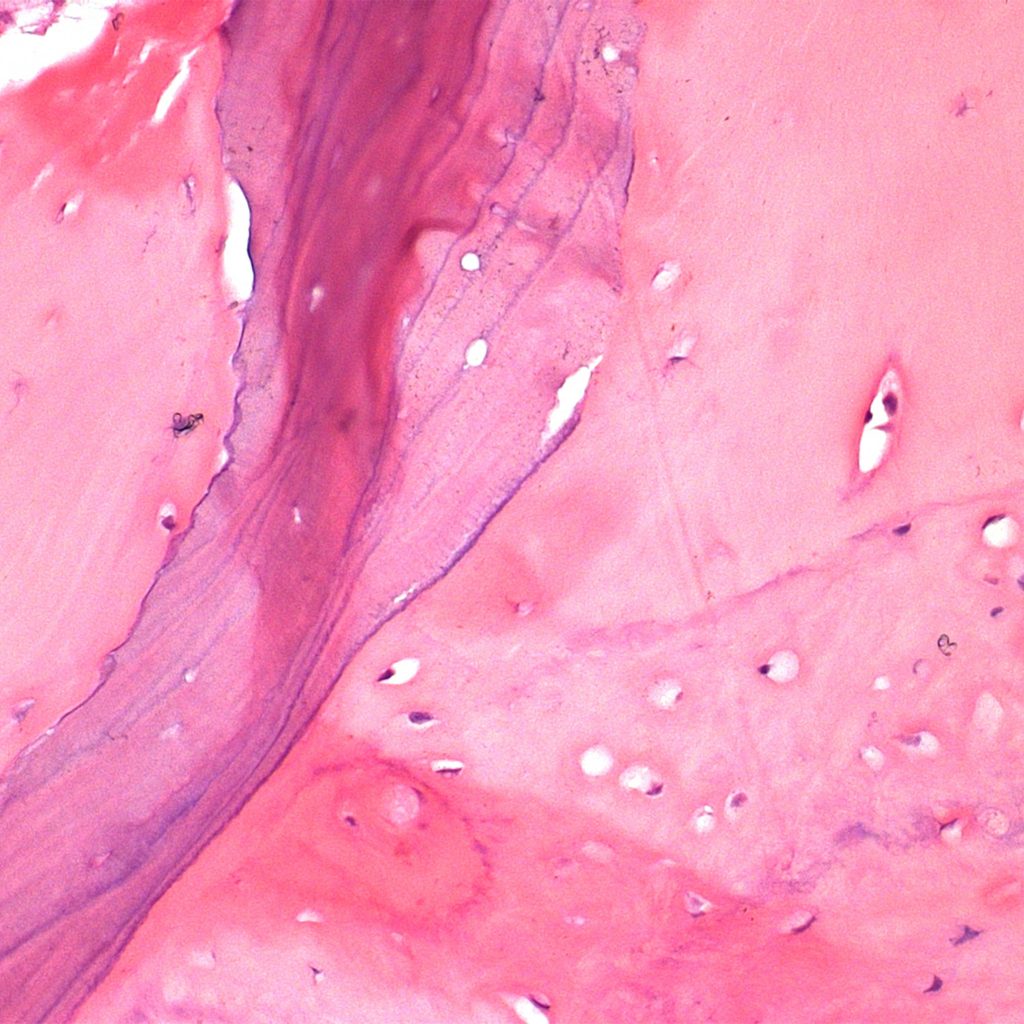
Histology.
Courtesy by. Dr Danilo Maeda I6 months sinus floor augmentation
Courtesy by. Dr Fábio Mizutani I 6 months sinus floor augmentation
Courtesy by. Dr Danilo Maeda I 6 months split crest
Indications.
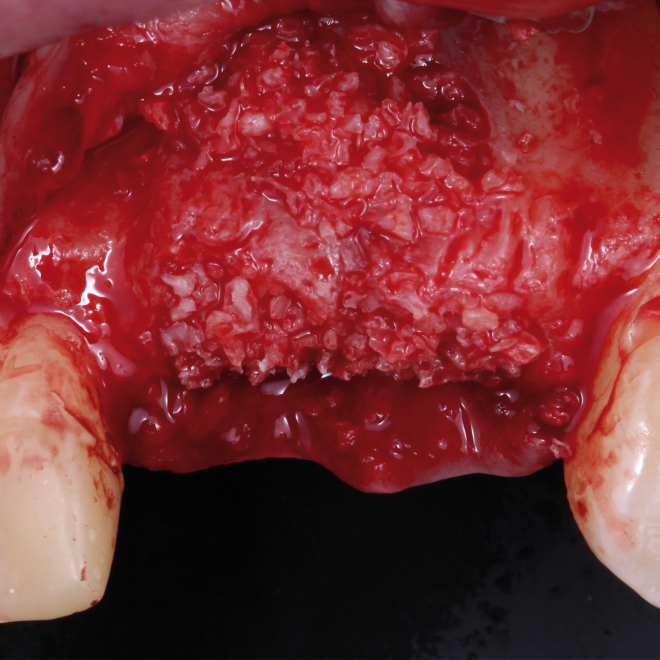
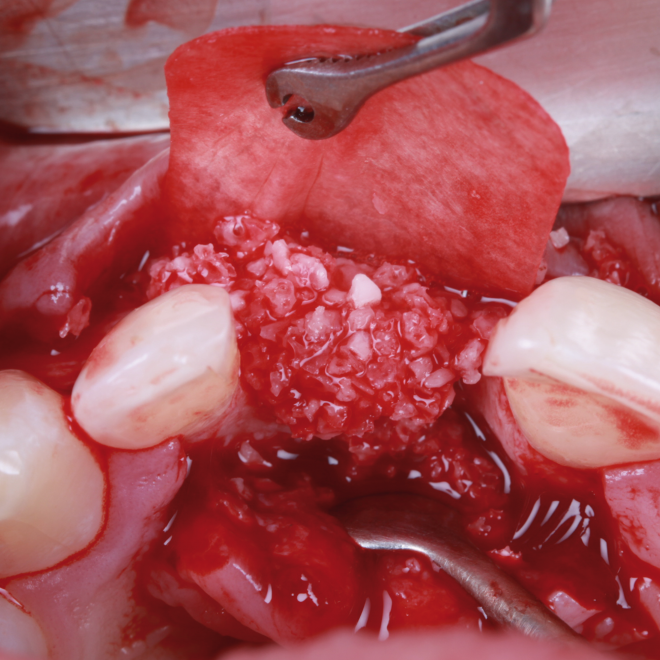
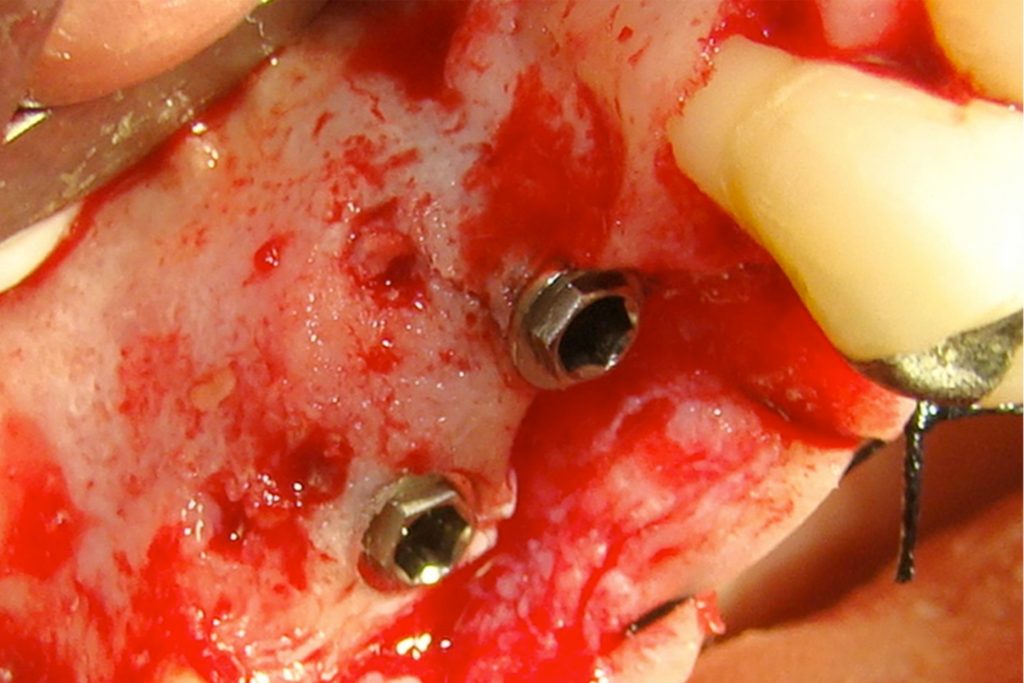
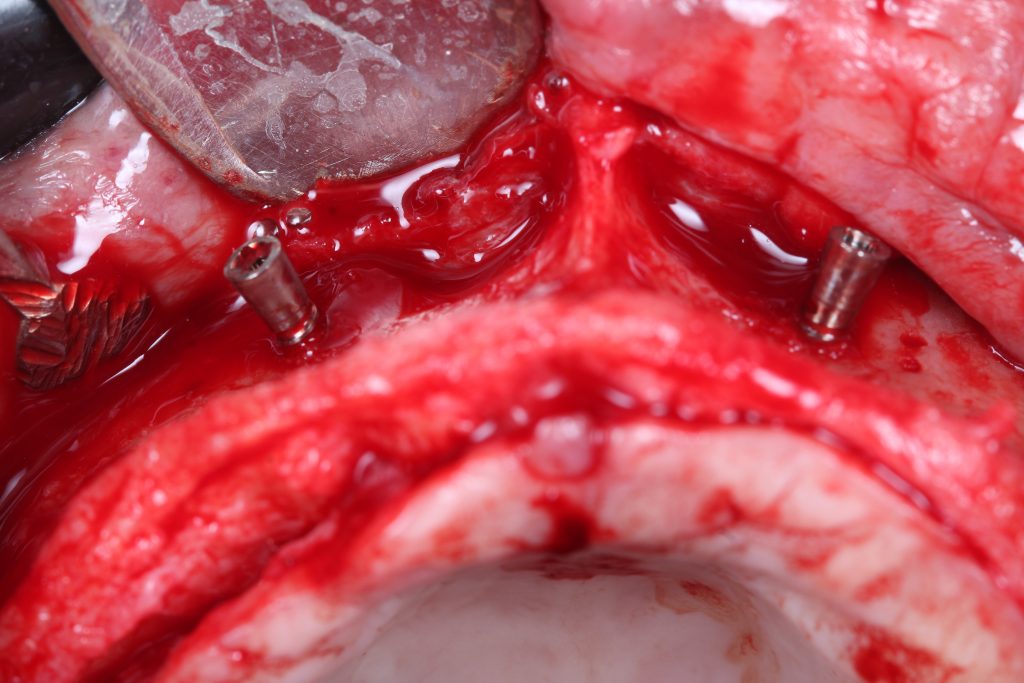
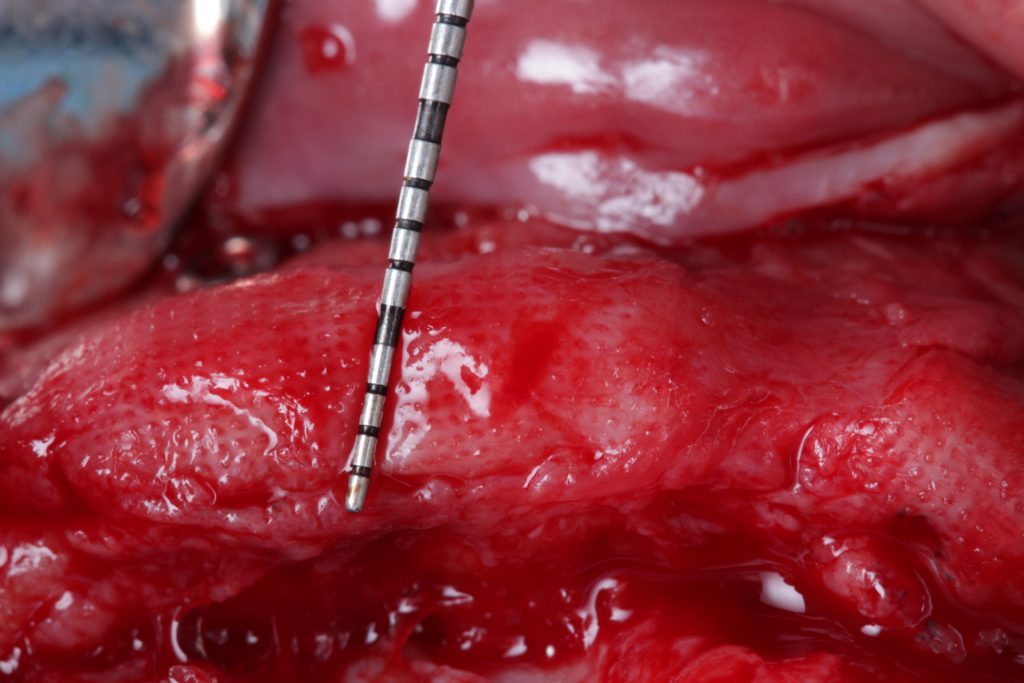
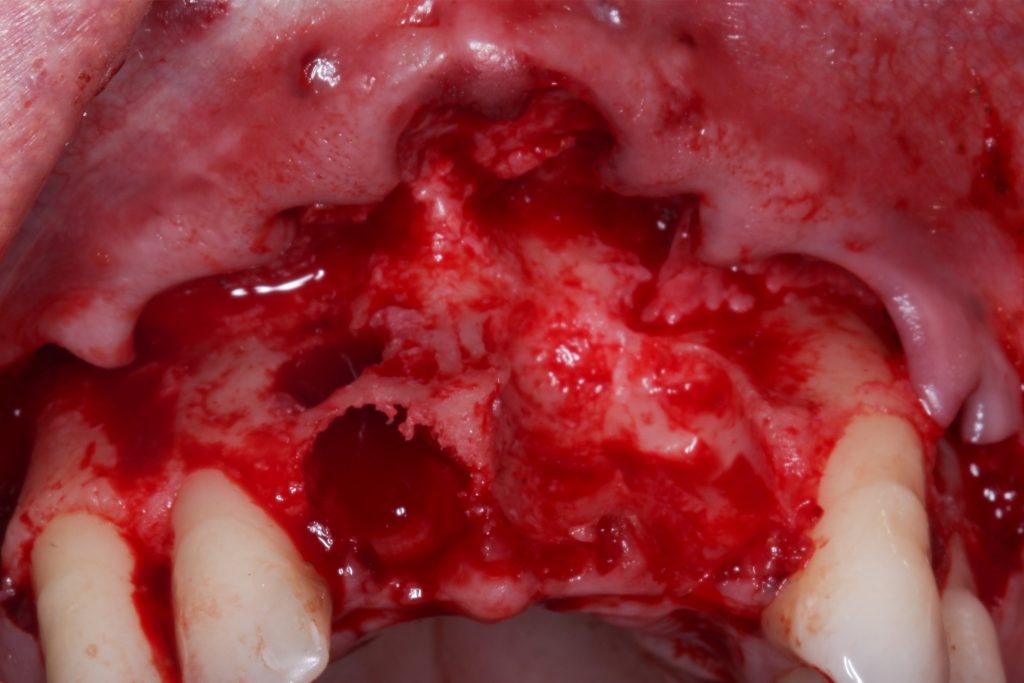
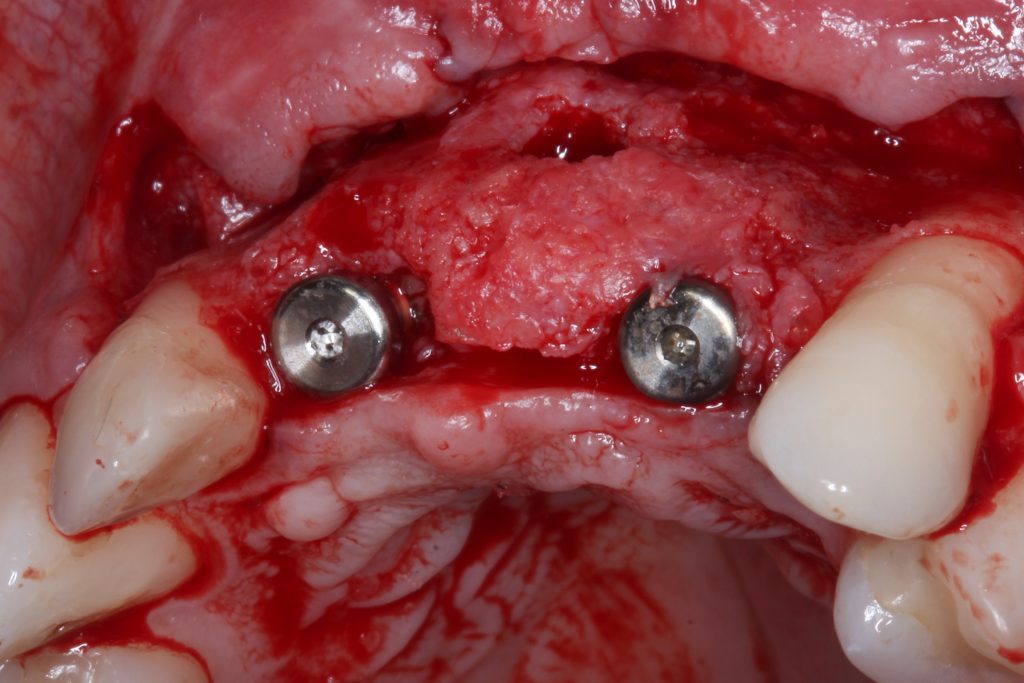
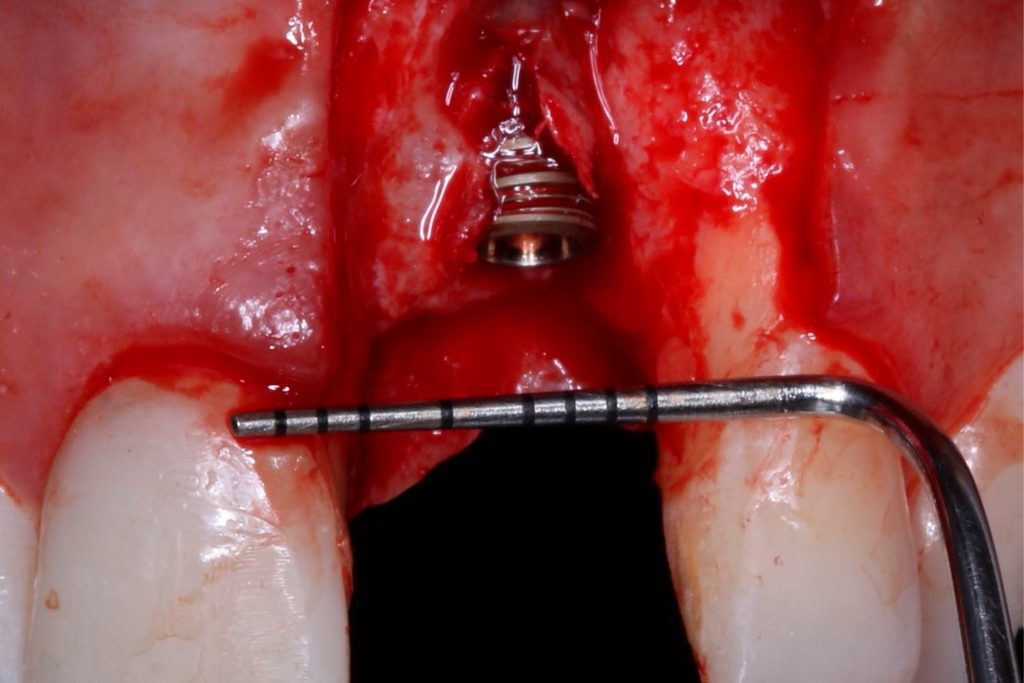
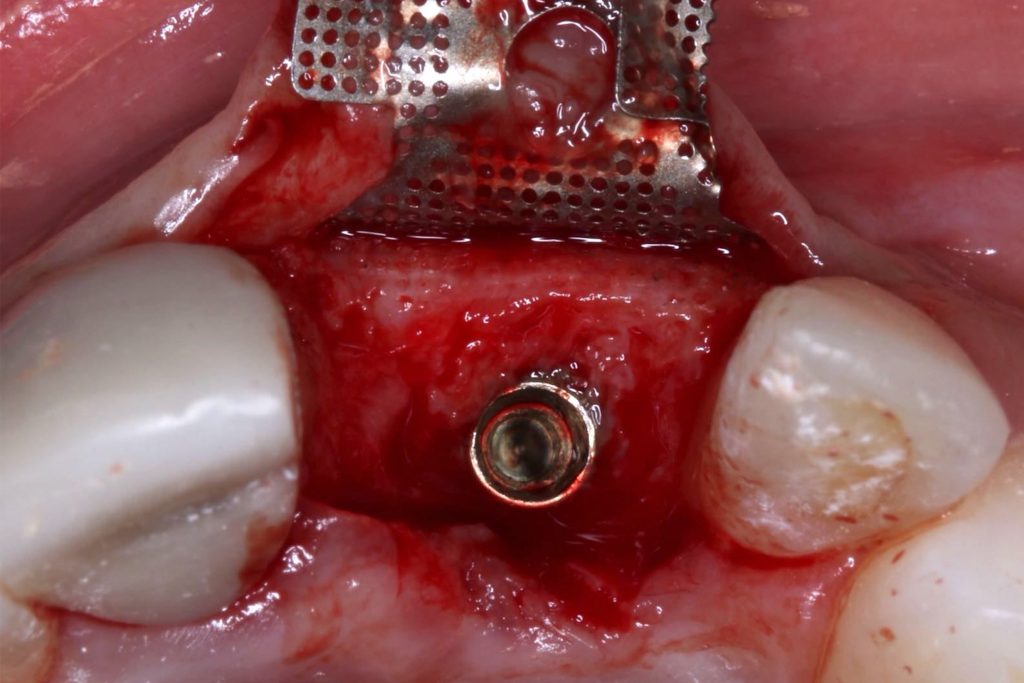
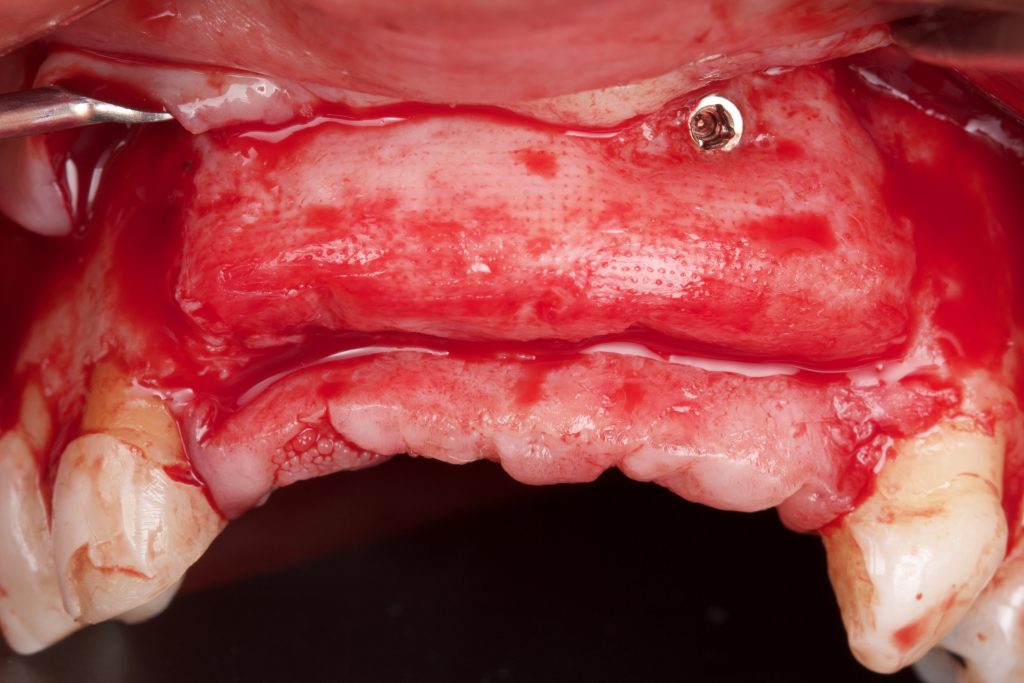
Bonefill is recommended for filling bone defects and for volumetric increase in the following situations: increase/reconstructions of alveolar crests, filling of post-extraction cavities, filling of cavities produced by post-surgery treatment interventions of cysts, granulomas and other lytic, oral and maxillofacial and dental pathologies, preparation of implant and filling sites of bone dehiscence, besides bone grafts in maxillary sinuses and in the periodontal area it can be used in filling bone defects and to support the membrane during guided bone regeneration.
Properties.
. Material Origin: Femoral heads of cattle
. Physicochemical treatment: Chemically treated - Receiving a sequence of baths [3 chemical process steps]
. Appearance Dry state: Whitish up to beige
. Structure Density: Controlled Inorganic particles of bovine matrix with high macro and micro porosity having a large inner surface area.
. Shelf life: 5 years
Clinical
Applications.




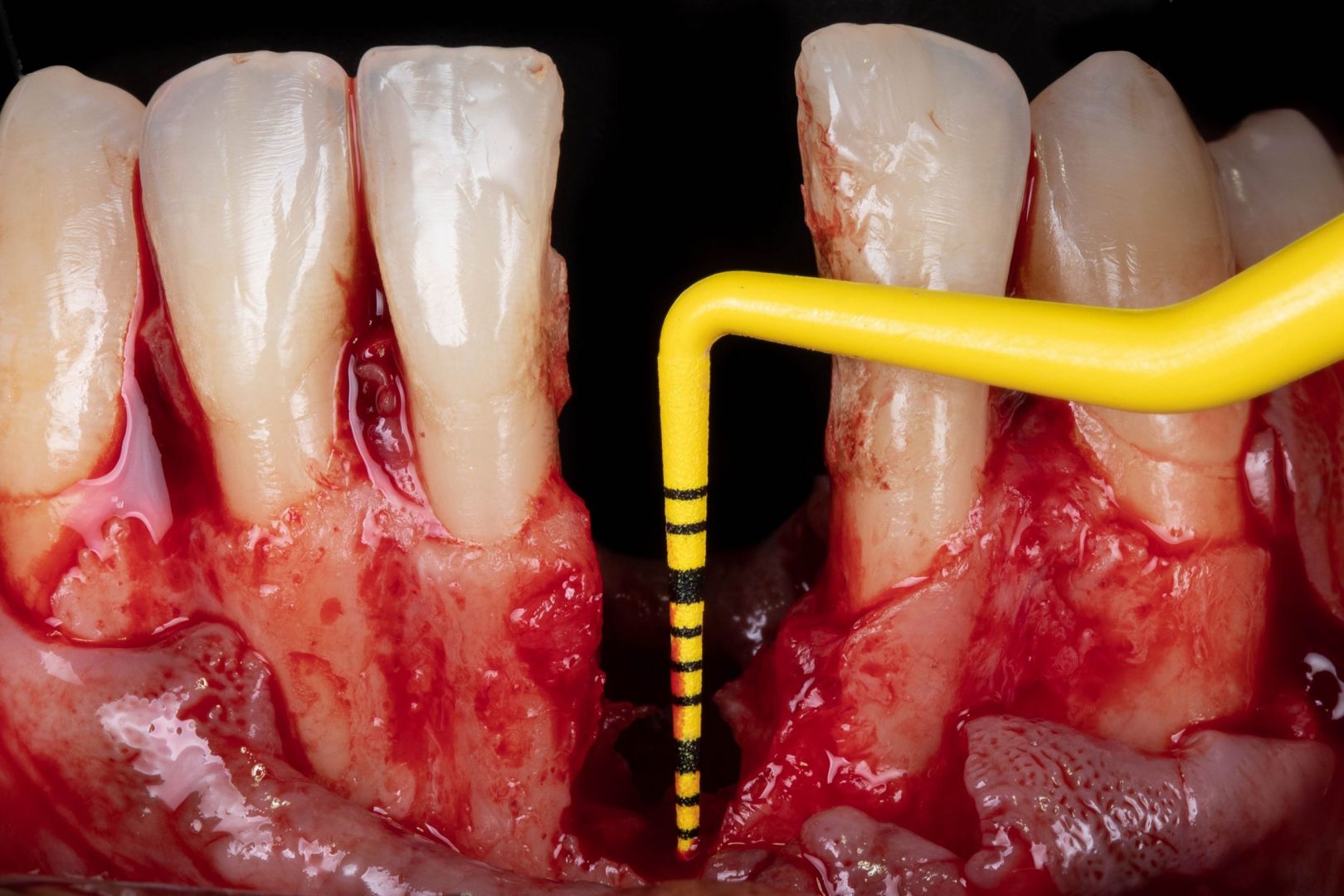
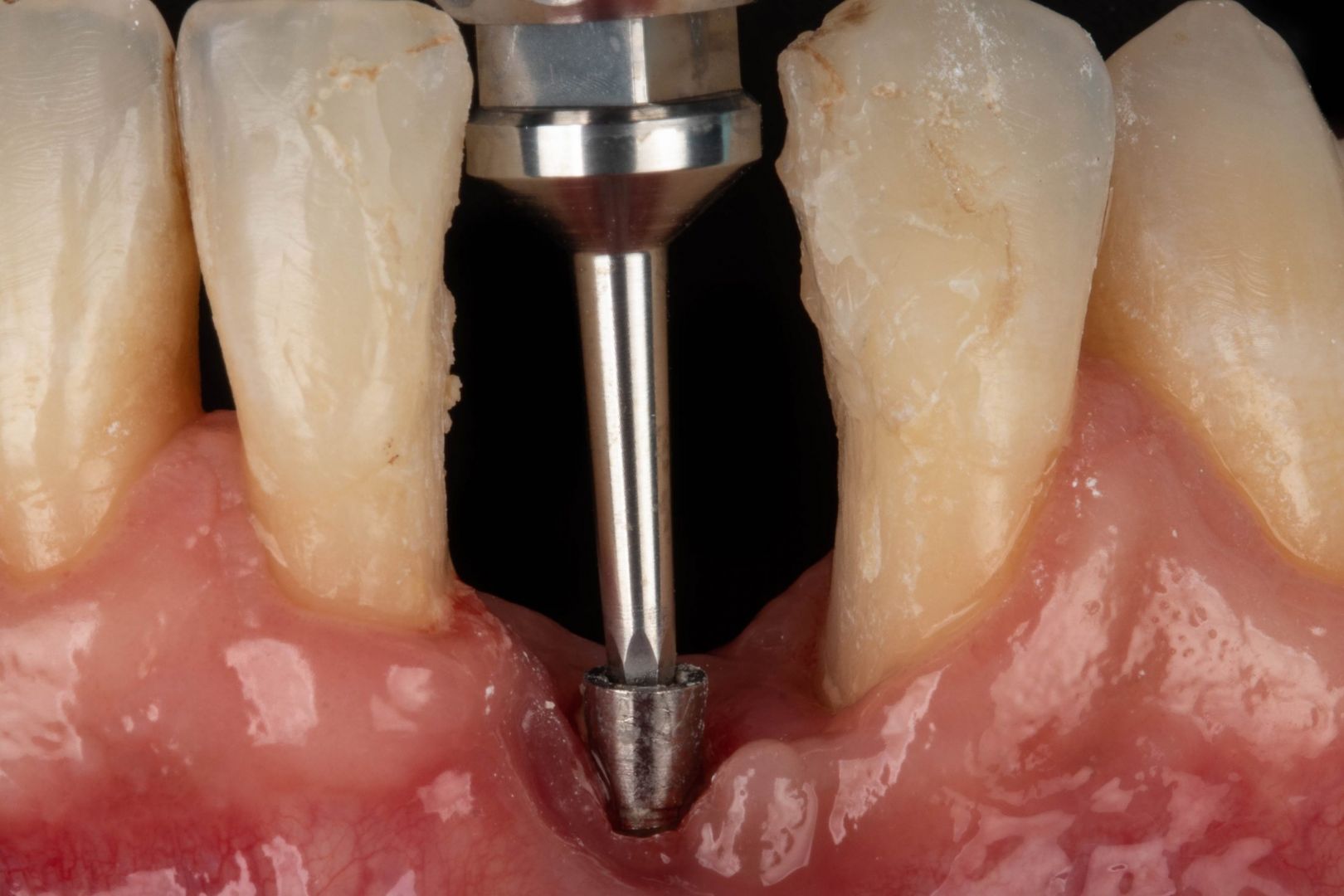
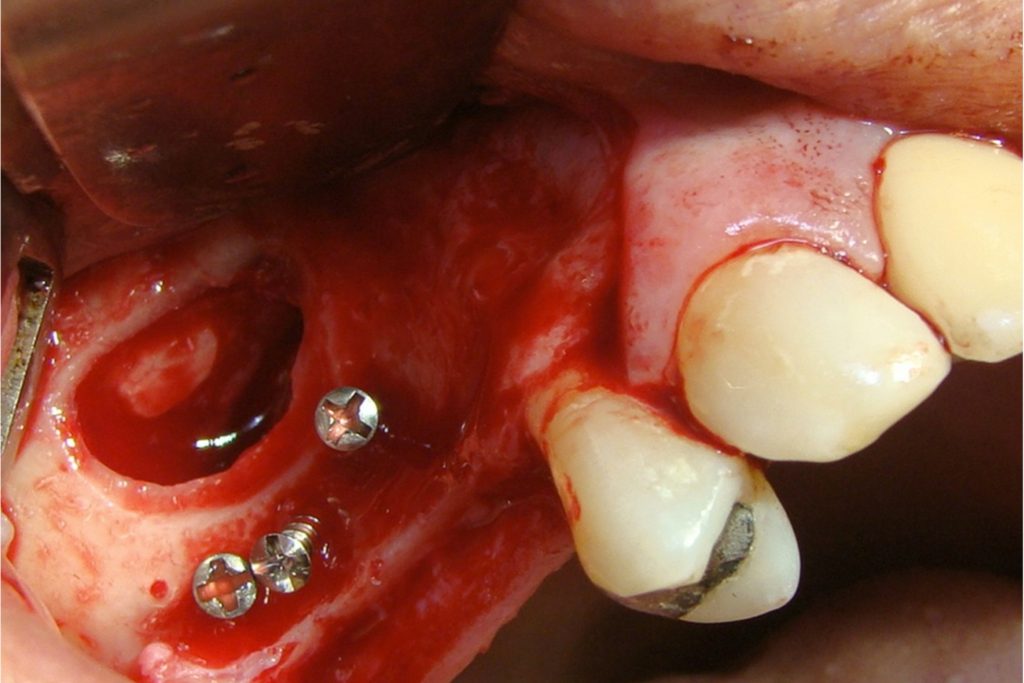

Videos.
Products
Specifications.
16001. Bonefill Denso [0,10- 0,60 mm] I Small 0,50g = 0,50cc
16024. Bonefill Denso [0,60- 1,50 mm] I Medium 0,50g = 0,50cc
16026. Bonefill Denso [1,50- 2,50 mm] I Large 0,50g = 1,00cc
16891. Bonefill Porous [0,10- 0,60 mm] I Small 1,0g = 1,50cc
16892. Bonefill Porous [0,60- 1,50 mm] I Medium 1,0g = 2,10cc
16893. Bonefill Porous [1,50- 2,50 mm] I Large 1,0g = 3,00cc
16955. Bonefill Mix [0,10- 1,50 mm] I Small/Medium 0,50g = 0,88cc
16964. Bonefill Mix [0,10- 1,50 mm] I Medium/Medium 0,50g = 0,88cc